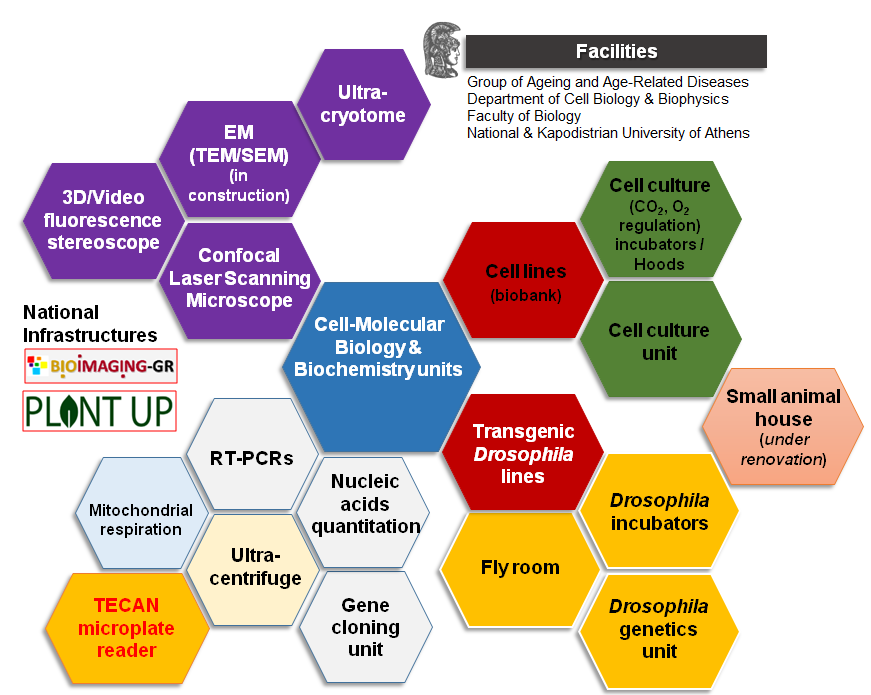
In terms of equipment and technological facilities our group is fully renovated and equipped with state-of-art research facilities including settings for molecular and cellular biology and biochemistry, as well as for insect and animal cells culturing including completely newly established cell culture and transgenic flies units, along with an animal house that hosts small scale studies on transgenic mice. Our group maintains a collection of numerous normal and cancer human/mice cell lines and of transgenic fly lines. In addition, our group has developed two mouse transgenic lines. Besides the above, our group members have at their disposal cutting edge facilities listed as national infrastructures (BIOIMAGING-GR, PLANT UP). Recent additions to the bioimaging unit are the upgrade of our confocal laser scanning microscope, the purchase of state-of-the-art equipment for cryo-electron microscopy sectioning, acquisition of a new inverted fluorescent scopes and a high resolution fluorescence stereoscope with high depth of field and maximum resolution for precisely detailed 3-D information. Last but not least, our lab owns a high throughput TECAN microplate reader, a hyper-centrifuge and a O2 concentration regulated cell culturing.
Bioimaging Unit (National Infrastructure partially funded by BIOIMAGING-GR):

Bioimaging Unit (National Infrastructure partially funded by BIOIMAGING-GR):

3D/Video fluorescence stereoscope: Leica M205 FA

Features:
20.5:1 motorized zoom with 7.8x-160x magnification range (1x objective, 10x eyepieces) and built-in digital display; Triple Beam™ technology- exclusive fluorescence illumination beam path for homogeneously illuminated, reflex-free field of view and excellent signal to noise ratio; FusionOptics™ - combines high resolution and depth of field, resulting in high fluorescence sensitivity and ideal optical 3-D impression; Resolution up to 525 lp/mm (1x objective, 10x eyepieces), maximum resolution up to 1050 lp/mm (2x objective, 10x eyepieces); Motorized and coded functions: zoom, four position fluorescence filter turret, iris diaphragm and objective nosepiece; Motorization and coding allows fast reproducibility and consistency in experimental procedures and permits calibrated measurement at all zoom levels; Fully apochromatic corrected optics allow high contrast, natural color images without color fringes or chromatic aberrations; The modular system offers complete flexibility and upgradability
Cryo-ultramicrotome: Leica EM FC6

Features:
20.5:1 motorized zoom with 7.8x-160x magnification range (1x objective, 10x eyepieces) and built-in digital display; Triple Beam™ technology- exclusive fluorescence illumination beam path for homogeneously illuminated, reflex-free field of view and excellent signal to noise ratio; FusionOptics™ - combines high resolution and depth of field, resulting in high fluorescence sensitivity and ideal optical 3-D impression; Resolution up to 525 lp/mm (1x objective, 10x eyepieces), maximum resolution up to 1050 lp/mm (2x objective, 10x eyepieces); Motorized and coded functions: zoom, four position fluorescence filter turret, iris diaphragm and objective nosepiece; Motorization and coding allows fast reproducibility and consistency in experimental procedures and permits calibrated measurement at all zoom levels; Fully apochromatic corrected optics allow high contrast, natural color images without color fringes or chromatic aberrations; The modular system offers complete flexibility and upgradability
Cryo-ultramicrotome: Leica EM FC6
The Leica EM FC6 is the low temperature sectioning system based on the Ultramicrotome Leica EM UC7 by mounting a Cryochamber. It provides preparation of semi- and ultrathin cryo-sections (-15° to -185°C) of biological and industrial samples for TEM, SEM, AFM and LM examination. For biological samples, The Leica EM FC6 is used for cryoprotected sectioning (Tokuyasutechniques) and Frozen Hydrated Sectioning (FHS). With the integrated antistatic device it provides good cryo-sectioning performance.
Ultra-cryotome: Leica AFS
The Leica EM AFS2 performs freeze substitution and progressive lowering of temperature (PLT) techniques and allows low temperature embedding and polymerization of resins. The Leica EM FSP (freeze substitution processor), an automatic reagent handling system combined with the Leica EM AFS2, dispenses reagents for both freeze substitution and PLT applications. The LED illumination from within the chamber and the attached stereomicroscope for viewing and positioning of samples ensures ease of use.
Cell- molecular biology and biochemistry units:


In vitro and in vivo experimental animal models units:

High-throughput screening unit (National Infrastructure partially funded by Plant UP-GR):

Features:
Absorbance (200 – 1000 nm); cuvette port; FI top / bottom;TRF, FRET; TR-FRET; FP; Luminescence flash & glow; Multi color luminescence (BRET1-3, NanoBRET); Ex/Em- fluorescence, absorbance, and lumi-scans; 2x Injectors; Integrated Gas Control; 1-1,535 well plates, NQ Plate; Cell ChipTM; Roboflask®; Te-CoolTM
Operation:
Fast absorbance scanning w. MCR; Fusion Optics for Fluorescence Intensity; Enhanced fluorescence (variable bandwidth; built-in and user-selectable dichroic mirrors; lens-based bottom reading); Luminescence scanning; Alpha Technology; FP with MCR; 3D-transport; Lid Lifter; Cell Counting / Image export; Automatic cell imaging; Microscope functionality; Cell confluence; Well Scanning; Automatic gain regulation for kinetic assays; Integrated barcode scanner; Integrated stacker - Spark-Stack
Absorbance (200 – 1000 nm); cuvette port; FI top / bottom;TRF, FRET; TR-FRET; FP; Luminescence flash & glow; Multi color luminescence (BRET1-3, NanoBRET); Ex/Em- fluorescence, absorbance, and lumi-scans; 2x Injectors; Integrated Gas Control; 1-1,535 well plates, NQ Plate; Cell ChipTM; Roboflask®; Te-CoolTM
Operation:
Fast absorbance scanning w. MCR; Fusion Optics for Fluorescence Intensity; Enhanced fluorescence (variable bandwidth; built-in and user-selectable dichroic mirrors; lens-based bottom reading); Luminescence scanning; Alpha Technology; FP with MCR; 3D-transport; Lid Lifter; Cell Counting / Image export; Automatic cell imaging; Microscope functionality; Cell confluence; Well Scanning; Automatic gain regulation for kinetic assays; Integrated barcode scanner; Integrated stacker - Spark-Stack
