We employ a multidisciplinary systems biology (cell-based, in vivo models) approach in order to understand:
a. the different levels of regulation and crosstalk of the various proteostatic machineries
b. the interaction, wiring and functional integration of proteostatic modules with mitostasis, metabolic pathways and genomic integrity
c. the systemic effects induced at the whole organism level by tissue-specific loss of proteostasis; and,
d. the deregulation of proteostatic and mitostatic modules in ageing and/or age-related diseases (e.g., cancer).
We are also actively involved in high-throughput screening for the identification of small molecules with anti-ageing and/or anti-cancer activity, as well in the study of the cellular-molecular effects induced by therapeutic anti-tumor proteasome inhibitors.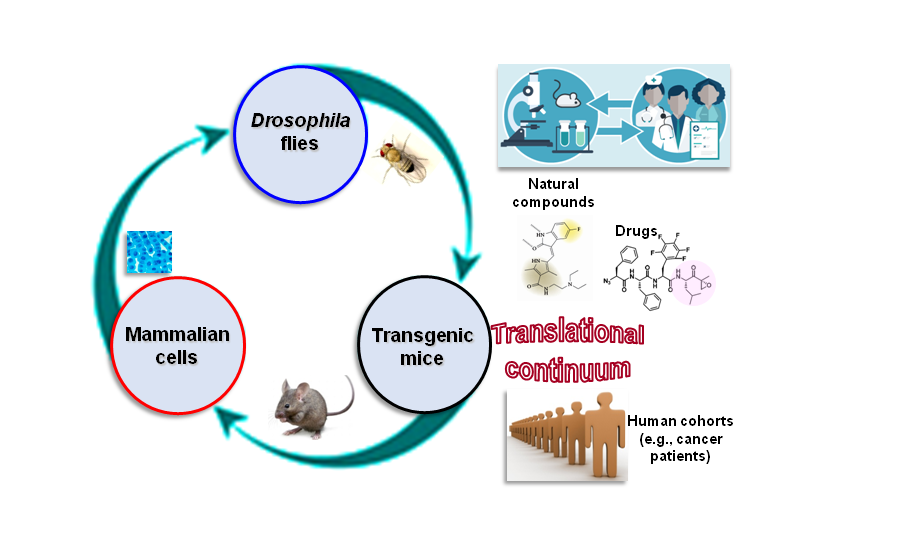
Proteins carry out the majority of cellular functions and maintain cellular homeodynamics mostly by participating in multimeric assemblies that operate as protein machines or complex 3D structures. Proteome quality control is thus critical for cellular function, and it is carried out through the curating activity of the proteostasis network (PN). Key components of the PN are the protein synthesis and trafficking modules, the endoplasmic reticulum unfolded protein response, molecular chaperones, and the two main degradation machineries, namely the ubiquitin-proteasome and autophagy-lysosome pathways (see also below). Moreover, central to the PN is the functional crosstalk of proteostasis and mitostasis that ensures mitochondrial quality control along with the interaction of proteostatic modules with major stress-responsive elements including nuclear factor erythroid 2-related factor 2 (Nrf2), which mobilizes genomic responses against oxidative and xenobiotic damage. The gradual accumulation of stressors during aging or earlier due to lifestyle results in an increasingly damaged and/or unstable proteome. Thus, deregulation of proteostasis is associated with several age-related diseases, and it is considered a major risk factor for a wide spectrum of protein conformational diseases, including immunological and metabolic disorders, neurodegenerative diseases and cancer. Reportedly, genetic or dietary interventions that activate components of the PN can delay aging in evolutionarily diverse organisms. Natural products (extracts or pure compounds) represent an extraordinary inventory of highly diverse structural scaffolds that offer promising activities towards meeting the challenge of increasing healthspan and/or delaying aging.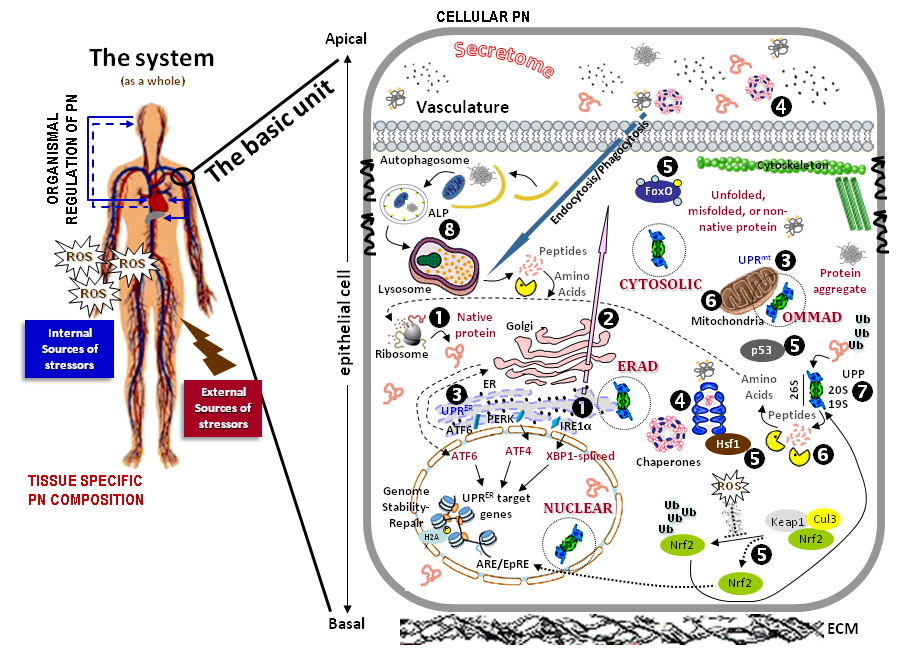
Overview of proteostatic machineries: Complex multicellular biological systems are an assembly of highly integrated subsystems, which are composed of a basic functional unit. Cells are also composed of smaller systems, which consist of smaller highly integrated systems. The PN is regulated at the organismal and tissue-specific levels and, during aging it is compromised by the accumulation of internally and externally originating stressors that promote the stochastic damage of all cellular biomolecules. Central to cell defense pathways is a multi-step regulatory process, in which PN plays a crucial role. Key components of the PN are the cytosolic and endoplasmic reticulum (ER) sites of protein synthesis (1), the modules of polypeptides sorting and trafficking (2), the unfolded protein response (UPR) of the ER and of the mitochondria (3), the intra- and extra-cellular molecular chaperones (4), as well as a number of transcription factors that function as stress sensors (Nrf2, Foxo, p53 or Hsf1) and mobilize genomic cytoprotective responses (5). Also, part of the PN are the compartmentalized (e.g. cytosolic or mitochondrial) (6) proteases, along with the ubiquitin-proteasome (UPP) (7) and autophagy-lysosome (ALP) (8) pathways. UPR-ER (3) consists of three types of ER stress sensors, namely the IRE1, PERK and ATF6 transcription factors that induce the expression of several proteins in order to restore ER proteostasis. Misfolded proteins that cannot be repaired are tagged with ubiquitin and are degraded mainly by the UPP. ROS production is a major source of cellular proteome damage and results in oxidative stress. ROS induce the activation of the Nrf2/Keap1 (5) signaling pathway, which regulates the expression of phase-II and antioxidant enzymes, as well as of proteasomal subunits in order to restore proteostasis. When the repairing/folding system of HSPs or the degrading network of proteases is overwhelmed, an extensive deterioration of cellular organelles occurs, which then prompts the activation of autophagic responses. Autophagy (8) starts with the formation of the autophagosome; subsequently, the membrane expands and eventually engulfs the cargo, closes its ends, and fuses with lysosomes in order to degrade its content. All these proteostatic modules are highly integrated by extensive functional crosstalk (from, Sklirou et al., Cancer Lett. 2018 Jan 28;413:110-121).
Selected recent publications:
Terpos E, Gavriatopoulou M, Ntanasis-Stathopoulos I, Briasoulis A, Gumeni S, Malandrakis P, Papanagnou ED, Migkou M, Kanellias N, Kastritis E, Trougakos IP, Dimopoulos MA. Booster BNT162b2 Optimizes SARS-CoV-2 Humoral Response in Myeloma Patients; the Negative Effect of anti-BCMA therapy. Blood. 2022 Jan 5:blood.2021014989. doi: 10.1182/blood.2021014989.
Manola MS, Gumeni S, Trougakos IP. Differential Dose- and Tissue-Dependent Effects of foxo on Aging, Metabolic and Proteostatic Pathways. Cells. 2021 Dec 18;10(12):3577. doi: 10.3390/cells10123577.
Bagratuni T, Markou A, Patseas D, Mavrianou-Koutsoukou N, Aktypi F, Liacos CI, Sklirou AD, Theodorakakou F, Ntanasis-Stathopoulos I, Gavriatopoulou M, Trougakos IP, Lianidou E, Terpos E, Kastritis E, Dimopoulos MA. Determination of MYD88L265P mutation fraction in IgM monoclonal gammopathies. Blood Adv. 2022 Jan 11;6(1):189-199. doi: 10.1182/bloodadvances.2021005354.
Gavriatopoulou M, Terpos E, Ntanasis-Stathopoulos I, Briasoulis A, Gumeni S, Malandrakis P, Fotiou D, Migkou M, Theodorakakou F, Eleutherakis-Papaiakovou E, Kanellias N, Kastritis E, Trougakos IP, Dimopoulos MA. Poor neutralizing antibody responses in 106 patients with WM after vaccination against SARS-CoV-2: a prospective study. Blood Adv. 2021 Nov 9;5(21):4398-4405. doi: 10.1182/bloodadvances.2021005444.
Neophytou CM, Trougakos IP, Erin N, Papageorgis P. Apoptosis Deregulation and the Development of Cancer Multi-Drug Resistance. Cancers (Basel). 2021 Aug 28;13(17):4363. doi: 10.3390/cancers13174363. PMID: 34503172 Free PMC article. Review.
Avgeris M, Adamopoulos PG, Galani A, Xagorari M, Gourgiotis D, Trougakos IP, Voulgaris N, Dimopoulos MA, Thomaidis NS, Scorilas A. Novel Nested-Seq Approach for SARS-CoV-2 Real-Time Epidemiology and In-Depth Mutational Profiling in Wastewater. Int J Mol Sci. 2021 Aug 7;22(16):8498. doi: 10.3390/ijms22168498. PMID: 34445204 Free PMC article.
Trougakos IP, Terpos E, Zirou C, Sklirou AD, Apostolakou F, Gumeni S, Charitaki I, Papanagnou ED, Bagratuni T, Liacos CI, Scorilas A, Korompoki E, Papassotiriou I, Kastritis E, Dimopoulos MA. Comparative kinetics of SARS-CoV-2 anti-spike protein RBD IgGs and neutralizing antibodies in convalescent and naïve recipients of the BNT162b2 mRNA vaccine versus COVID-19 patients.BMC Med. 2021 Aug 23;19(1):208. doi: 10.1186/s12916-021-02090-6.
Bergamaschi C, Terpos E, Rosati M, Angel M, Bear J, Stellas D, Karaliota S, Apostolakou F, Bagratuni T, Patseas D, Gumeni S, Trougakos IP, Dimopoulos MA, Felber BK, Pavlakis GN. Systemic IL-15, IFN-γ, and IP-10/CXCL10 signature associated with effective immune response to SARS-CoV-2 in BNT162b2 mRNA vaccine recipients. Cell Rep. 2021 Aug 10;36(6):109504. doi: 10.1016/j.celrep.2021.109504. Epub 2021 Jul 23. PMID: 34352226
Terpos E, Gavriatopoulou M, Ntanasis-Stathopoulos I, Briasoulis A, Gumeni S, Malandrakis P, Fotiou D, Papanagnou ED, Migkou M, Theodorakakou F, Roussou M, Eleutherakis-Papaiakovou E, Kanellias N, Trougakos IP, Kastritis E, Dimopoulos MA. The neutralizing antibody response post COVID-19 vaccination in patients with myeloma is highly dependent on the type of anti-myeloma treatment. Blood Cancer J. 2021 Aug 2;11(8):138. doi: 10.1038/s41408-021-00530-3. PMID: 34341335
Gumeni S, Papanagnou ED, Manola MS, Trougakos IP. Nrf2 activation induces mitophagy and reverses Parkin/Pink1 knock down-mediated neuronal and muscle degeneration phenotypes. Cell Death Dis. 2021 Jul 3;12(7):671. doi: 10.1038/s41419-021-03952-w. PMID: 34218254 Free PMC article.
Tsakiri EN, Gumeni S, Manola MS, Trougakos IP. Amyloid toxicity in a Drosophila Alzheimer's model is ameliorated by autophagy activation. Neurobiol Aging. 2021 Sep;105:137-147. doi: 10.1016/j.neurobiolaging.2021.04.017.
Terpos E, Trougakos IP, Gavriatopoulou M, Papassotiriou I, Sklirou AD, Ntanasis-Stathopoulos I, Papanagnou ED, Fotiou D, Kastritis E, Dimopoulos MA. Low neutralizing antibody responses against SARS-CoV-2 in older patients with myeloma after the first BNT162b2 vaccine dose. Blood. 2021 Jul 1;137(26):3674-3676. doi: 10.1182/blood.2021011904. PMID: 33861315 Free PMC article. No abstract available.
Terpos E, Trougakos IP, Apostolakou F, Charitaki I, Sklirou AD, Mavrianou N, Papanagnou ED, Liacos CI, Gumeni S, Rentziou G, Korompoki E, Papassotiriou I, Dimopoulos MA. Age-dependent and gender-dependent antibody responses against SARS-CoV-2 in health workers and octogenarians after vaccination with the BNT162b2 mRNA vaccine. Am J Hematol. 2021 Jul 1;96(7):E257-E259. doi: 10.1002/ajh.26185.
Cheimonidi C, Grivas IN, Sesti F, Kavrochorianou N, Gianniou DD, Taoufik E, Badounas F, Papassideri I, Rizzi F, Tsitsilonis OE, Haralambous S, Trougakos IP. Clusterin overexpression in mice exacerbates diabetic phenotypes but suppresses tumor progression in a mouse melanoma model. Aging (Albany NY). 2021 Mar 10;13(5):6485-6505. doi: 10.18632/aging.202788. Epub 2021 Mar 10. PMID: 33744871
Klionsky DJ, et al. Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition)1. Autophagy. 2021 Jan;17(1):1-382. doi: 10.1080/15548627.2020.1797280. Epub 2021 Feb 8.
Kastritis E, Laina A, Georgiopoulos G, Gavriatopoulou M, Papanagnou ED, Eleutherakis-Papaiakovou E, Fotiou D, Kanellias N, Dialoupi I, Makris N, Manios E, Migkou M, Roussou M, Kotsopoulou M, Stellos K, Terpos E, Trougakos IP, Stamatelopoulos K, Dimopoulos MA. Carfilzomib-induced endothelial dysfunction, recovery of proteasome activity, and prediction of cardiovascular complications: a prospective study. Leukemia. 2021 May;35(5):1418-1427. doi: 10.1038/s41375-021-01141-4.
Trougakos IP, Stamatelopoulos K, Terpos E, Tsitsilonis OE, Aivalioti E, Paraskevis D, Kastritis E, Pavlakis GN, Dimopoulos MA. Insights to SARS-CoV-2 life cycle, pathophysiology, and rationalized treatments that target COVID-19 clinical complications. J Biomed Sci. 2021 Jan 12;28(1):9. doi: 10.1186/s12929-020-00703-5. PMID: 33435929 Free PMC article.
Gkogkou E, Barnasas G, Vougas K, Trougakos IP. Expression profiling meta-analysis of ACE2 and TMPRSS2, the putative anti-inflammatory receptor and priming protease of SARS-CoV-2 in human cells, and identification of putative modulators. Redox Biol. 2020 Sep;36:101615. doi: 10.1016/j.redox.2020.101615. Epub 2020 Jun 24. PMID: 32863223 Free PMC article.
Sarmento-Ribeiro AB, Scorilas A, Gonçalves AC, Efferth T, Trougakos IP. The emergence of drug resistance to targeted cancer therapies: Clinical evidence. Drug Resist Updat. 2019 Dec;47:100646. doi: 10.1016/j.drup.2019.100646. Epub 2019 Sep 26. PMID: 31733611
Kalioraki MA, Artemaki PI, Sklirou AD, Kontos CK, Adamopoulos PG, Papadopoulos IN, Trougakos IP, Scorilas A. Heat shock protein beta 3 (HSPB3) is an unfavorable molecular biomarker in colorectal adenocarcinoma. Mol Carcinog. 2020 Jan;59(1):116-125. doi: 10.1002/mc.23133. Epub 2019 Nov 10. PMID: 31709619
Skorda A, Sklirou AD, Sakellaropoulos T, Gianniou DD, Kastritis E, Terpos E, Tsitsilonis OE, Florea BI, Overkleeft HS, Dimopoulos MA, Alexopoulos LG, Trougakos IP. Non-lethal proteasome inhibition activates pro-tumorigenic pathways in multiple myeloma cells. J Cell Mol Med. 2019 Dec;23(12):8010-8018. doi: 10.1111/jcmm.14653. Epub 2019 Sep 30. PMID: 31568628 Free PMC article.
Alexa-Stratulat T, Pešić M, Gašparović AČ, Trougakos IP, Riganti C. What sustains the multidrug resistance phenotype beyond ABC efflux transporters? Looking beyond the tip of the iceberg. Drug Resist Updat. 2019 Sep;46:100643. doi: 10.1016/j.drup.2019.100643. Epub 2019 Aug 23. PMID: 31493711 Free article.
Trougakos IP. Nrf2, stress and aging. Aging (Albany NY). 2019 Aug 2;11(15):5289-5291. doi: 10.18632/aging.102143. Epub 2019 Aug 2. PMID: 31377746 Free PMC article.
Functional wiring of proteostatic and mitostatic modules ensures transient organismal survival during imbalanced mitochondrial dynamics. Gumeni S, Evangelakou Z, Tsakiri EN, Scorrano L, Trougakos IP. Redox Biol. 2019 Jun;24:101219. doi: 10.1016/j.redox.2019.101219.
Tsakiri EN, Gumeni S, Vougas K, Pendin D, Papassideri I, Daga A, Gorgoulis V, Juhász G, Scorrano L, Trougakos IP. Proteasome dysfunction induces excessive proteome instability and loss of mitostasis that can be mitigated by enhancing mitochondrial fusion or autophagy. Autophagy. 2019 Oct;15(10):1757-1773. doi: 10.1080/15548627.2019.1596477.
Tsakiri EN, Gumeni S, Iliaki KK, Benaki D, Vougas K, Sykiotis GP, Gorgoulis VG, Mikros E, Scorrano L, Trougakos IP. Hyperactivation of Nrf2 increases stress tolerance at the cost of aging acceleration due to metabolic deregulation. Aging Cell. 2019 Feb;18(1):e12845. doi: 10.1111/acel.12845.
Efentakis P, Kremastiotis G, Varela A, Nikolaou PE, Papanagnou ED, Davos CH, Tsoumani M, Agrogiannis G, Konstantinidou A, Kastritis E, Kanaki Z, Iliodromitis EK, Klinakis A, Dimopoulos MA, Trougakos IP, Andreadou I, Terpos E. Molecular mechanisms of carfilzomib-induced cardiotoxicity in mice and the emerging cardioprotective role of metformin. Blood. 2019 Feb 14;133(7):710-723. doi: 10.1182/blood-2018-06-858415.
Gorgoulis VG, Pefani DE, Pateras IS, Trougakos IP. Integrating the DNA damage and protein stress responses during cancer development and treatment. J Pathol. 2018 Sep;246(1):12-40. doi: 10.1002/path.5097. Epub 2018 Jul 19. PMID: 29756349 Free PMC article.
Sklirou A, Papanagnou ED, Fokialakis N, Trougakos IP. Cancer chemoprevention via activation of proteostatic modules. Cancer Lett. 2018 Jan 28;413:110-121. doi: 10.1016/j.canlet.2017.10.034.
Tsakiri EN, Gaboriaud-Kolar N, Iliaki KK, Tchoumtchoua J, Papanagnou ED, Chatzigeorgiou S, Tallas KD, Mikros E, Halabalaki M, Skaltsounis AL, Trougakos IP. The Indirubin Derivative 6-Bromoindirubin-3'-Oxime Activates Proteostatic Modules, Reprograms Cellular Bioenergetic Pathways, and Exerts Antiaging Effects. Antioxid Redox Signal. 2017 Nov 10;27(14):1027-1047. doi: 10.1089/ars.2016.6910.
